 The Guide facilitates the effective and efficient use of valuable resources by the application of appropriate and proportionate practices, encouraging innovative approaches to managing risk to patient safety, product quality, and data integrity, while supporting benefit to public health.Through the principles and methodologies suggested here, this guide will help the organization to ensure that computer systems prove their fitness for intended use, meet the good practices of the industry in an efficient manner, provide practical guidance to facilitate the interpretation of regulatory requirements, with a language and terminology easy to understand and interpret, clarify the roles and responsibilities of each of those involved in the validation of computerized systems.įinally, this guide is designed for understanding the principles of validation of computerized systems by the most diverse personnel, both those who occupy this knowledge as part of their daily work and those who at some point will be involved in the effort to validate a system without any prior knowledge of Good Practices, validation or IT computer terminology, thus becomes a valuable tool for both and for anyone who wants to train others in basic and logical principles of work on Computer Systems Validation.Ĭurrently, the Health Industries such as pharmaceuticals (human and veterinary), pharmochemical, (APIs and excipients), biologics, biotechnology, blood products, and medical devices, are required to establish a validation program to demonstrate that any procedure, process, equipment, material, activity or system actually leads to the expected results. 
Technological innovation is essential for life sciences industries in providing value to society while also controlling costs and reducing time to market. 
The ISPE GAMP ® 5 Guide: A Risk-Based Approach to Compliant GxP Computerized Systems Second Edition aims to protect patient safety, product quality, and data integrity by facilitating and encouraging the achievement of computerized systems that are effective, reliable, and of high quality. GAMP ® guidance does not define a prescriptive method or a standard, but rather provides pragmatic guidance, approaches, and tools for the practitioner. 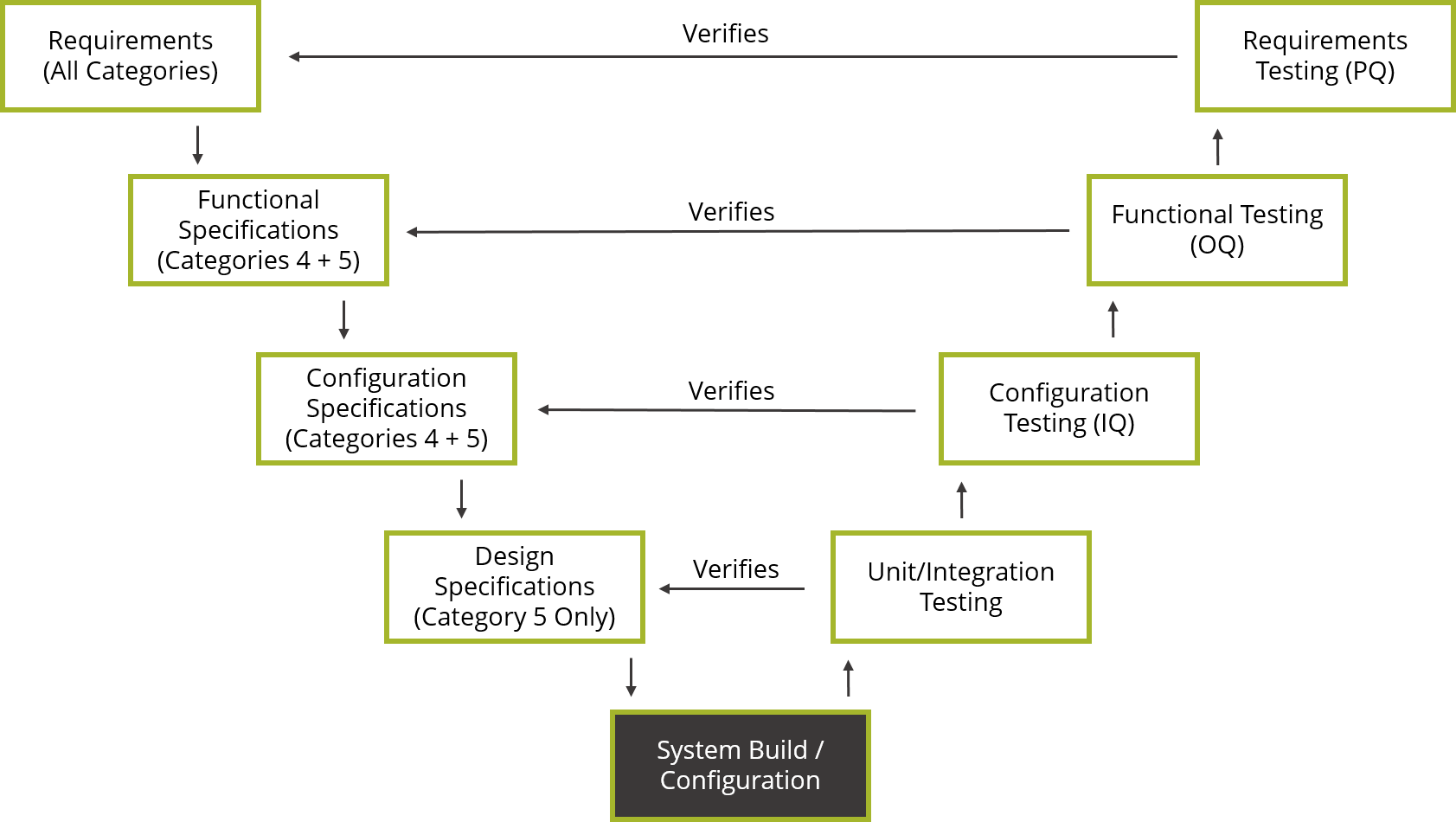
Promotes a system life cycle approach based on good practice.Establishes a common language and terminology.Facilitates the interpretation of regulatory requirements.GAMP® is an ISPE Community of Practice (CoP). 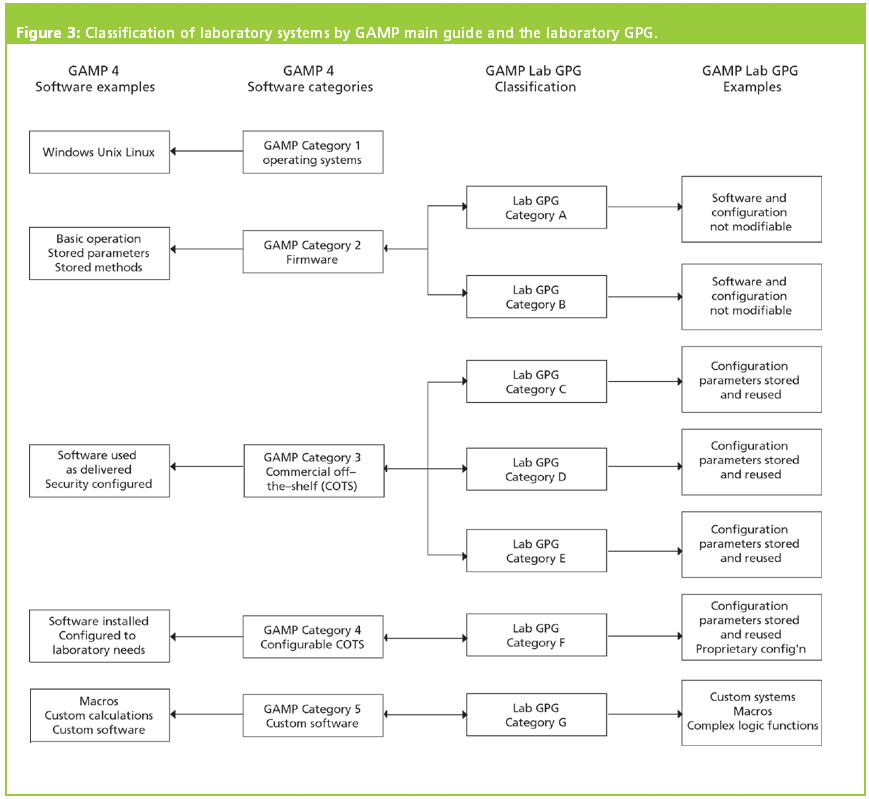
GAMP® adopts a patient-centric risk-based approach that enables innovation while demonstrating compliance with regulatory requirements. It aims to achieve computerized systems that are fit for intended use and meet current regulatory requirements by building upon existing industry good practice in an efficient and effective manner. GAMP ® guidance aims to safeguard patient safety, product quality, and data integrity in the use of GxP computerized systems. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
